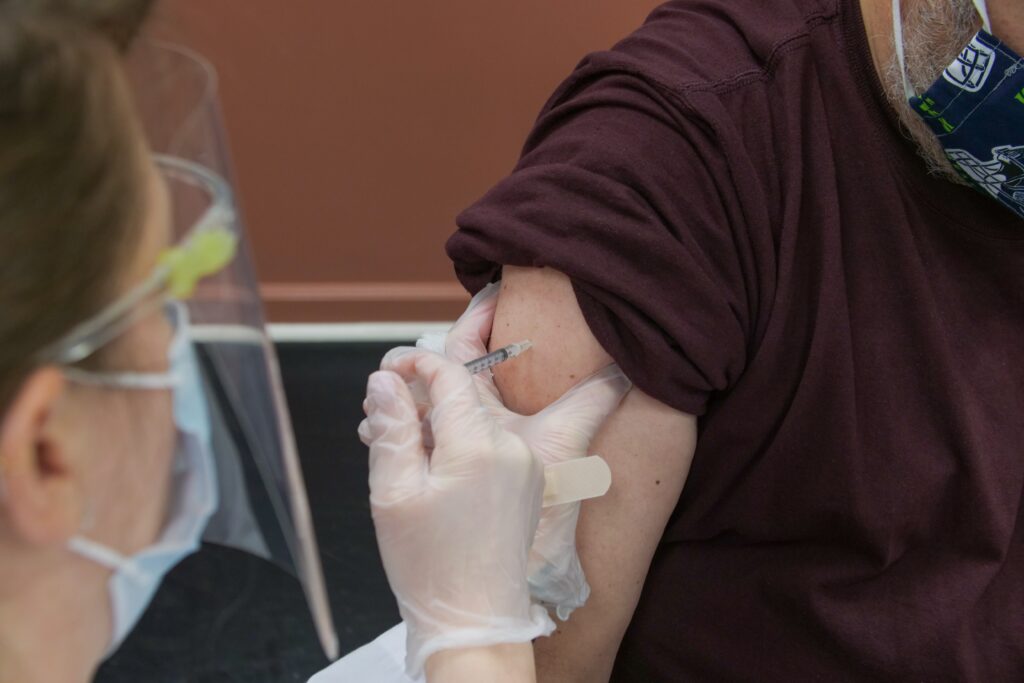
In response to the devastating impact of COVID-19, healthcare organizations have been hard at work to create a vaccine capable of addressing the risks brought by COVID-19. Now that the vaccine is approved, Caring Places Management has partnered with three local pharmacies to administer the vaccine to all residents that would like to take it.
A Push for Safety
At Caring Places, we have assumed the responsibility of caring for your loved ones with safety and well-being as a leading priority of our care model. The CDC has listed residents in long term care facilities as priority recipients for the COVID-19 vaccine. As part of our commitment to protecting your loved one’s health, we have partnered with three local pharmacies, Walgreens, Consonsus, and Omnicare, to administer the vaccine to residents in all of our communities.
We strongly encourage all of our residents and staff to give consent to receiving the vaccine, as it is considered a lifesaving option against COVID-19. The vaccine has been rigorously tested and gone through numerous clinical trials before being authorized for use. We address some of the more common fears or questions we’ve received about the vaccine.
1. It’s been rushed to the market and isn’t safe. The FDA only authorizes the use of a vaccine after extensive, large-scale clinical trials that yielded significant amounts of reviewable data. After reviewing this data, the FDA ruled that the vaccine was appropriate for use.
2. The vaccine is mandatory. While highly recommended, this vaccine has not been declared a mandatory injection. Residents and staff must agree to participate and receive the vaccine.
3. The side effects are severe. Potential side-effects, if any, typically include pain at the injection site of vaccination, as well as possible headache or muscle aches. These usually last for no more than a day or two. However, compared to the severity of symptoms with COVID-19, the vaccinations’ side effects are more manageable and less dangerous.
The Plan for Our Communities
We hope that each staff and resident at all of our communities will sign the consent form to receive the vaccine. As a senior living community, we are included in the national rollout phase to administer the vaccine. Under the federal rollout, long-term care members receiving the COVID-19 vaccine are a priority. Each Caring Places facility will determine which pharmacy will be administering the vaccine.
Before an individual receives the vaccine, a consent form is required to be signed. For those who are unable to sign, we will contact the patient’s health care power of attorney to receive consent. Those who do not want to participate in the vaccination are not required to attend the vaccination clinics. The vaccine requires two doses, with a separation of 21-28 days between each injection, depending on which vaccine is administered. Side effects from the vaccine are similar to what may be experienced when receiving a flu shot, typically with mild discomfort only lasting a few days.
Our desire is for each member of our community, staff or resident, to get vaccinated. The more residents and staff we get vaccinated, the better we can protect our communities and reduce the chances of a COVID-19 outbreak. We will continue to follow the CDC recommended COVID-19 protocols after the vaccination clinics, adhering to social distancing recommendations, wearing personal protective equipment, and sanitizing our facilities.
The Promise of Our Care
Our efforts to administer COVID-19 vaccines to residents of long-term care facilities supports our overall commitment to ensure the health and well-being of your loved one. Our facilities are a supportive, safe environment for those who need a little extra help making it through their daily activities and those living with dementia and Alzheimer’s. Despite all that 2020 has brought, our promise of delivering the best care possible will not change. Let us know how we can help you transition your loved one into one of our Caring Places communities.